Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.
Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature.
Gas chromatography is the process of separating compounds in a mixture by injecting a gaseous or liquid sample into a mobile phase, typically called the carrier gas, and passing the gas through a stationary phase. The mobile phase is usually an inert gas or an unreactive gas such as helium, argon, nitrogen or hydrogen. The stationary phase is a microscopic layer of viscous liquid on a surface of solid particles on an inert solid support inside a piece of glass or metal tubing called a column. The surface of the solid particles may also act as the stationary phase in some columns. The glass or metal column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled and the eluent coming off the column is monitored by a computerized detector.
GC Analysis
A gas chromatograph is a chemical analysis instrument for separating chemicals in a complex sample. A gas chromatograph is made up of a narrow flow-through tube, known as the column, through which the sample passes in a gas stream (the carrier gas) at different rates depending on their various chemical and physical properties and their interaction with a specific column lining or filling, called the ‘stationary phase’. As the chemicals exit the end of the column, they are detected and identified electronically. The function of the stationary phase in the column is to separate different components, causing each one to exit the column at a different time. Other parameters that can be used to alter the order or time of retention are the carrier gas flow rate, column length and the temperature.
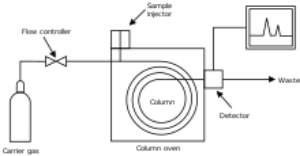
In a GC analysis, a known volume of gaseous or liquid analyte is injected through a rubber disk and into a hot, temperature controlled, port attached to the column. As the carrier gas transports the analyte molecules through the column, there is adsorption of the analyte molecules either onto the column walls or onto packing materials (stationary phase) in the column to give separation. Since each type of molecule has a different rate of progression, the various components of the analyte mixture are separated as they progress along the column and reach the end of the column at different times (retention time). A detector is used to monitor the time at which each component reaches the outlet and ultimately the amount of that component can be determined. Generally, substances are identified (qualitatively) by the order in which they elute from the column and by the retention time of the analyte in the column.
Physical components of a GC include;
1. Autosampler.
2. Inlets.
3. Detectors.
Applications
In general, substances that vaporize below 300 °C (and therefore are stable up to that temperature) can be measured quantitatively. The samples are also required to be salt-free; they should not contain ions. Very minute amounts of a substance can be measured, but it is often required that the sample must be measured in comparison to a sample containing the pure, suspected substance known as a reference standard.
Various temperature programs can be used to make the readings more meaningful; for example to differentiate between substances that behave similarly during the GC process.
Professionals working with GC analyze the content of a chemical product, for example in assuring the quality of products in the chemical industry; or measuring toxic substances in soil, air or water. GC is very accurate if used properly and can measure picomoles of a substance in a 1 ml liquid sample, or parts-per-billion concentrations in gaseous samples.
In practical courses at colleges, students sometimes get acquainted to the GC by studying the contents of Lavender oil or measuring the ethylene that is secreted by Nicotiana benthamiana plants after artificially injuring their leaves. These GC analyse hydrocarbons (C2-C40+).
In a typical experiment, a packed column is used to separate the light gases, which are then detected with a TCD (Thermal Conductivity Detectors). The hydrocarbons are separated using a capillary column and detected with a FID (Flame Ionization Detectors). A complication with light gas analyses that include H2 is that He, which is the most common and most sensitive inert carrier (sensitivity is proportional to molecular mass) has an almost identical thermal conductivity to hydrogen (it is the difference in thermal conductivity between two separate filaments in a Wheatstone Bridge type arrangement that shows when a component has been eluted). For this reason, dual TCD instruments used with a separate channel for hydrogen that uses nitrogen as a carrier are common. Argon is often used when analysing gas phase chemistry reactions such as F-T synthesis so that a single carrier gas can be used rather than two separate ones. The sensitivity is reduced, but this is a trade off for simplicity in the gas supply.
Gas chromatography is used extensively in forensic science. Disciplines as diverse as solid drug dose (pre-consumption form) identification and quantification, arson investigation, paint chip analysis, and toxicology cases, employ GC to identify and quantify various biological specimens and crime-scene evidence.
Reference: https://en.wikipedia.org/wiki/Gas_chromatography

