Gas chromatography (GC) is a separation technique used to isolate volatile components of a mixture depending on differences in the mode of partitioning between a flowing mobile phase and a stationary phase. Because of its simplicity, sensitivity, and effectiveness, GC has earned its place as one of the most important tools in chemistry. Columns/stationary phases are considered the “heart” or “brain” of the chromatograph and are responsible for the separation process. In the GC system, a sample is vaporized and injected into the head of the separation column packed with a finely divided solid or coated with a film of a liquid. When a sample traverses the column by the flow of an inert gas employed as the mobile phase, its components are separated owing to differences in their interactions with the stationary phase. Upon elution from the column, the separated compounds pass over a detector that generates a signal corresponding to the concentration of the compound. The species present can be qualitatively identified based on the delay in the sample passing through the column. Extensive research has led to improved columns for achieving better separation and resolution. Since the initial development of packed columns, many technological advancements have been made. The capillary column was the first advancement in which stationary phases fabricated by using the latest technology was employed.
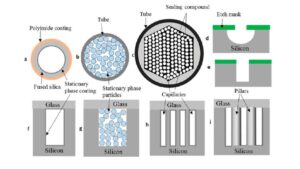
Fig 1: Cross sectional view of different GC-columns.
Main GC Modes:
GC is the only form of chromatography that does not utilize a mobile phase for interacting with the analyte. When the stationary phase is a solid adsorbent, the process is termed gas–solid chromatography (GSC), and when it is a liquid on an inert support, the process is termed gas–liquid chromatography (GLC)
Main GC Columns:
In GLC, the liquid stationary phase is adsorbed onto a solid inert packing or immobilized on the capillary tubing walls. The column is considered packed if the glass or metal column tubing is filled with small spherical inert supports. The liquid phase is adsorbed onto the surface of these beads in a thin layer. In a capillary column, the tubing walls are coated with the stationary phase or an adsorbent layer that is able to support the liquid phase. However, GSC has a limited application in the laboratory and is rarely used due to severe peak tailing and semipermanent retention of polar compounds within the column. Therefore, the term “gas–liquid chromatography” is simply shortened to “gas chromatography.”
Packed GC columns:
Because the first commercial instruments accepted only packed columns, all initial studies of GC were performed on packed columns. Packed columns are typically made of stainless steel and have an outer diameter of 0.64 or 0.32 cm and lengths of 0.61–3.05 m. Alternative inert materials have also been used, including glass, nickel, fluorocarbon polymer (Teflon), and steel covered with glass or Teflon. The packing is an inert support impregnated with 5–20% stationary phase. The solid support holds the liquid stationary phase and should have a large surface area (0.5–5 m2 /g), be chemically inert, have low sorptive activity toward common analytes, and have good mechanical strength to prevent the fracture of the coated particles during loading and handling. Diatomaceous earth, composed of hydrous silica with impurities, has been used as a solid support under the brand name Chromosorb. The surfaces of the diatomaceous earth support have many active sites generated from free hydroxyl groups that form undesirable hydrogen bonds with polar solute molecules and cause peak tailing. Even the most inert material (white Chromosorb) must be washed with acid and silanized to make it more inert. Some typical deactivated white supports are Supelcoport, Chromosorb WHP, Gas Chrom Q II, and Anachrom Q. The disadvantage of deactivation is that these supports become hydrophobic, and coating them with a polar stationary liquid can be difficult. GC liquid phases are primarily silicone-based oils with high temperature stability. These liquid phases are available in a range of polarities, extending from methyl silicone (OV-1, OV-101, SE-30, and DC-200), methyl phenyl silicone (OV-17 and SE-52), and methyl trifluoropropyl silicone (OV-210 and QF-1) to methyl cyanoethyl silicone (OV-225 and AN-600). The use of a higher liquid phase load (∼10%) both decreases the adsorptive interactions between the solute and the solid support and increases the column capacity to prevent overloading from dirty samples, but requires the use of a higher column temperature for elution.
Capillary GC Columns:
Although capillary columns were introduced in 1959, they did not gain popularity until 1980. At present, it is estimated that more than 80% of all applications are run on capillary columns due to the fast and efficient separation the afford. Capillary chromatographic columns are not filled with packing material; instead, a thin film of liquid phase coats the inner wall. Because the tube is open, its resistance to flow is very low, and it is thus referred to as an open tubular column. Open tubular columns can be divided into three groups and are described in the next sections.
- Porous Layer Open Tubular Column: Porous layer open tubular (PLOT) columns, first suggested by Golay in the late 1950s, have been successfully developed and commercialized. PLOT columns contain a porous layer of a solid adsorbent such as alumina, molecular sieves, or Porapak. PLOT columns are well suited for the analysis of light, fixed gases, and other volatile compounds.
- Wall-Coated Open Tubular Column: In 1957, Golay demonstrated the superiority of wall-coated open tubular (WCOT) columns (a 100-fold or higher increase in efficiency) relative to packed columns; yet, it took a quarter century before this efficiency was realized in practice. In WCOT columns, the wall is directly coated with the stationary-phase layer at a film thickness of 0.05–3 μm.
- Support-Coated Open Tubular Column: Support-coated open tubular (SCOT) columns were introduced in 1963 by Halász and Horváth. These columns contain an adsorbed layer of a very fine solid support (such as Celite) coated with the liquid phase. SCOT columns can hold more liquid phase and have a higher sample capacity than the thin films of early wall-coated open tubular (WCOT) columns had. With the introduction of cross-linking techniques, the use of stable, thick films in WCOT columns has become possible, thereby making SCOT columns redundant.
Fused Silica Open Tubular Column:
Stainless steel was utilized early as a material for capillary GC. However, due to the lack of efficiency and high reactivity with compounds, including steroids, amines, and free acids, the steel capillary has been outdated. Glass columns suffer from the drawback of being fragile. Thus, fused silica was introduced in 1979, and almost all capillary columns are made of fused silica. The fused silica tubes have much thinner walls than glass capillary columns, and are strengthened by the polyimide coating. These columns are flexible and can be wound into coils. They offer the advantages of physical strength, flexibility, and low reactivity.

Fig 2: Fused silica capillary GC columns.
Reference:
https://onlinelibrary.wiley.com/doi/full/10.1002/9783527678129.assep024
